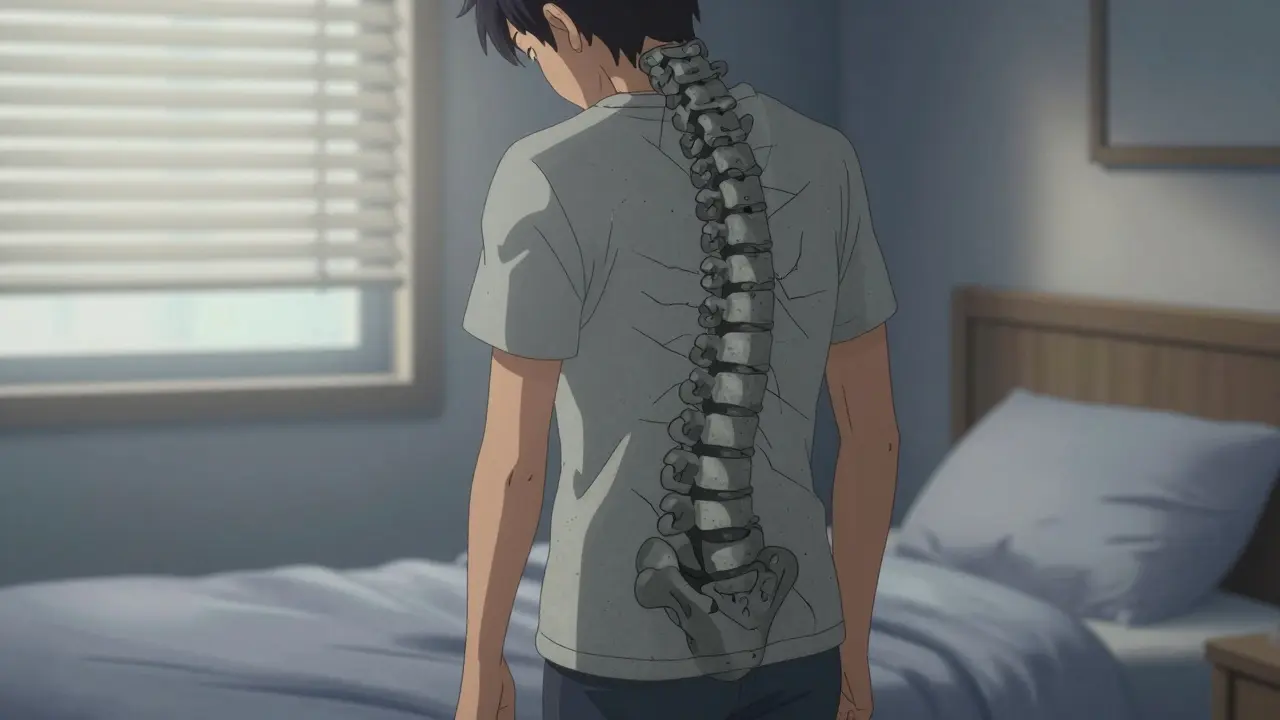
Imagine waking up every morning feeling like your spine has been fused into a solid block of concrete. For people living with Ankylosing Spondylitis is a chronic inflammatory rheumatic disease that primarily attacks the axial skeleton, leading to progressive spinal fusion and joint damage. It isn't just a "bad back"; it's a systemic battle where your own immune system attacks the joints and ligaments, potentially robbing you of your mobility if left unchecked. But there is a silver lining: the arrival of biological therapies has fundamentally changed the trajectory of this disease.
The Mechanics of Spine Inflammation
To understand how to treat the condition, we have to look at what's actually happening inside the body. The process typically moves through three brutal phases: inflammation, bone erosion, and finally, new bone formation. This progression often starts in the sacroiliac joints is the set of joints connecting the lower spine to the pelvis. When these joints become inflamed, the body tries to heal itself, but in AS, this healing process goes haywire, creating bony bridges that eventually lock the spine in place.
A huge piece of the puzzle is the HLA-B27 is a specific genetic marker or protein found on the surface of white blood cells. While not everyone with this gene gets the disease, about 5-6% of those who are positive for it will develop AS. If you're experiencing chronic morning stiffness that lasts over an hour and improves with movement but not with rest, your body might be signaling an inflammatory surge.
What are TNF Inhibitors?
If your immune system is a fire, then Tumor Necrosis Factor-alpha (TNF-α) is a pro-inflammatory cytokine, or signaling protein, that acts as the primary fuel for the inflammatory cascade in AS. It's particularly abundant in the SI joints and is the main driver behind the early, painful stages of the disease.
TNF inhibitors are biological drugs designed to seek out and neutralize this protein. By blocking TNF-α, these medications stop the "fire" from spreading, which reduces pain, lowers stiffness, and can potentially slow down the radiographic progression of the disease. They aren't like standard painkillers; they target the biological root of the inflammation rather than just masking the symptoms.
Comparing the Top TNF Inhibitor Options
Not all biologics are created equal. Depending on your lifestyle, how you prefer to take medication, and your specific symptoms, one might be a better fit than others. Some are given as a drip in a clinic, while others are simple injections you do at home.
| Drug Name | Administration | Frequency | Key Characteristic |
|---|---|---|---|
| Infliximab | Intravenous (IV) | Every 4-8 weeks | Administered in a clinical setting |
| Etanercept | Subcutaneous | Twice weekly | Often shows longest drug persistence |
| Adalimumab | Subcutaneous | Every other week | Widely used; available as biosimilars |
| Certolizumab pegol | Subcutaneous | Weekly or Bi-weekly | Pegylated structure for longer half-life |
| Golimumab | Subcutaneous | Monthly | Lowest injection frequency |

When Should You Start Biologic Therapy?
You don't usually jump straight to biologics. The standard path starts with nonsteroidal anti-inflammatory drugs (NSAIDs). However, if you've used maximum tolerated doses of NSAIDs for four weeks and your spinal pain is still high, it's time to talk about a transition. Doctors typically use the BASDAI is the Bath Ankylosing Spondylitis Disease Activity Index, a patient-reported tool to measure disease severity to decide. A score of 4 or higher, combined with persistent pain, usually triggers a recommendation for a TNF inhibitor.
Predicting who will respond best is an art and a science. Research shows that patients who are younger, have a shorter disease duration, and have elevated inflammatory markers like C-reactive protein (CRP) tend to see the most dramatic improvements. In fact, when both CRP and serum amyloid A are high at the start, there's an 81% chance of a positive outcome.
Real-World Results and Side Effects
The impact of these drugs is often life-changing. In clinical trials, about 58-65% of patients achieve an ASAS20 response (a 20% improvement in disease activity). In the real world, this translates to shorter morning stiffness-often dropping from over an hour to under 30 minutes-and a return to activities like gardening, sports, or simply sitting comfortably in a chair.
But these drugs aren't without risks. Because they dampen your immune system, you're more prone to infections. This is why doctors insist on screening for tuberculosis and hepatitis before the first dose. Some users also report skin reactions or the development of psoriasis. For instance, some patients might find great relief with etanercept but have to switch to adalimumab if a skin condition flares up. It's a balancing act, but for the majority, the trade-off is worth the regain of mobility.

Practical Tips for Starting Treatment
If you're moving toward self-injection, don't let the needle intimidate you. Most people get the hang of it within a few supervised sessions. The learning curve is short, and by the fourth week, over 90% of patients are successfully managing their own doses.
- Screening First: Ensure you have your Quantiferon or tuberculin skin test completed to rule out latent TB.
- Track Your Response: Keep a daily log of your morning stiffness duration to see if the drug is working.
- Rotation is Key: When injecting subcutaneously, rotate your injection sites (thigh, abdomen) to avoid skin irritation.
- Stay Vigilant: Report any unusual fever or persistent cough to your rheumatologist immediately, as your immune response is suppressed.
The Future of AS Management
We are entering an era of personalized medicine. We're moving beyond a one-size-fits-all approach and looking at HLA-B27 subtypes to predict exactly which drug will work for which person. There's also exciting research into more selective inhibitors that block only the "bad" pro-inflammatory receptors while leaving the "protective" ones alone.
While new players like interleukin-17 inhibitors (such as secukinumab) are entering the market and showing great results, TNF inhibitors remain the gold standard due to twenty years of safety data. If caught early-ideally within two years of symptom onset-these therapies can reduce radiographic progression by up to 60%, effectively stopping the "fusion" before it becomes permanent.
Do TNF inhibitors cure Ankylosing Spondylitis?
No, they do not cure AS because it is a chronic genetic condition. However, they can lead to clinical remission, where symptoms disappear and inflammation is controlled, preventing further spinal fusion.
Can I switch from one TNF inhibitor to another?
Yes. If the first drug loses efficacy or causes side effects, about 50-60% of patients are candidates for a second TNF inhibitor. Roughly 30-40% of those who switch find a successful second option.
How long does it take for these drugs to start working?
Many patients report a noticeable improvement within the first 2-3 doses. However, the full therapeutic benefit typically takes up to 12 weeks to manifest completely.
What is the difference between a biologic and a biosimilar?
A biologic is the original patented drug. A biosimilar is a highly similar version created after the patent expires. Biosimilars, like Amjevita, provide the same clinical benefits but are often 15-20% cheaper.
Are there any long-term risks of cancer with TNF inhibitors?
Recent data from the British Society for Rheumatology suggests there is no increased malignancy risk compared to the general AS population, with a standardized incidence ratio very close to 1.0.







Joel Bonstell
May 1, 2026 AT 12:47Man, the morning stiffness is the worst part of this whole thing. I've been on a biologic for a few years now and it really does make a huge difference, but you gotta be patient with the first few doses. Just keep at it and don't let the needles scare you off, they're way easier than they look!
bharat films
May 2, 2026 AT 21:39Typical pharma propaganda pushing expensive biologics 🙄 The ROI on these drugs is basically zero for the patient since the cost is insane and the side effects are just shifted around. Absolute joke 🤡🤡
Mikaela -anonymous 😏
May 2, 2026 AT 22:54Oh wow... a chart!!! How revolutionary!!!! I'm just so deeply moved by the bravery of people taking shots in their thighs... truly a spiritual journey... 🙄🙄🙄
Kelly Feehely
May 4, 2026 AT 02:26Wake up people!! You think the "Big Pharma" companies just happen to have these "biosimilars" ready when the patent expires? It's all a controlled market cycle to keep us dependent on synthetic proteins that mess with our natural homeostasis. They don't want you cured, they want a subscription model for your immune system! Absolute garbage!
Jenny X
May 4, 2026 AT 23:59The systemic modulation of cytokine activity is precisely where the cognitive dissonance occurs in the general public. If you analyze the pharmacokinetics of TNF-alpha inhibitors, it becomes evident that the epigenetic markers are being manipulated via exogenous proteins. It's a classic case of synthetic intervention masking a deeper biochemical imbalance designed by the industrial complex to ensure lifelong dependency.
Spencer Farrell
May 5, 2026 AT 07:06One must consider that the pursuit of pharmaceutical relief is often a surrogate for the failure of the individual to harmonize their existence with the biological imperatives of the human form. While the clinical data presented is ostensibly accurate, it lacks a metaphysical understanding of inflammation as a manifestation of internal discord. It is a tragedy of the modern age that we prioritize the suppression of a protein over the cultivation of the self.
Kartik Agarwal
May 6, 2026 AT 21:26For anyone struggling with the transition from NSAIDs, remember that the titration period can be challenging. The BASDAI score is a great metric, but don't overlook the importance of a multidisciplinary approach involving physiotherapy to maintain joint articulation while the biologics handle the systemic inflammation. You've got this!
kelvin villa saab
May 7, 2026 AT 12:11I lapped the doctor last week and he totaly missed the mark on my CRP levels. Most of these docs just want to put you on the first drug they see in the manual without actually lookin at the patient. Its a disgraece to the medical profesion to be this lazy with diagnosis!!
princess lovearies
May 8, 2026 AT 15:55It's okay to feel overwhelmed by the diagnosis. Just take it one day at a time and remember that your value isn't defined by your mobility. We are all just doing our best to navigate these bodies.
Allison Maier
May 10, 2026 AT 15:39Too many words. Just take the shot or dont. 😒
Jimmy Crocker
May 10, 2026 AT 18:51I find the anecdotal evidence regarding the efficacy of biosimilars to be somewhat pedestrian at best, though I suppose for the average layman the distinction between a reference product and its biosimilar counterpart is merely a matter of fiscal convenience, despite the subtle nuances in glycosylation patterns that might, in a more rigorous clinical setting, reveal a disparate immunogenic profile that most clinicians simply ignore in their haste to optimize billing codes.
Seema Karanje
May 11, 2026 AT 13:08STOP COMPLAINING AND START MOVING! If you have AS, you can't afford to be lazy! Get your meds, do your exercises, and FIGHT BACK against this disease every single day! No excuses!
Elizabeth Holden
May 13, 2026 AT 10:53lol these drugs are just overpriced salt water. My cousin took one and he felt fine for a week then he got a cold and it was all over. The data is fake and the doctors are just lying to make money. basic science 101 people!!